Clinical Trials
Your Partner for clinical supply management
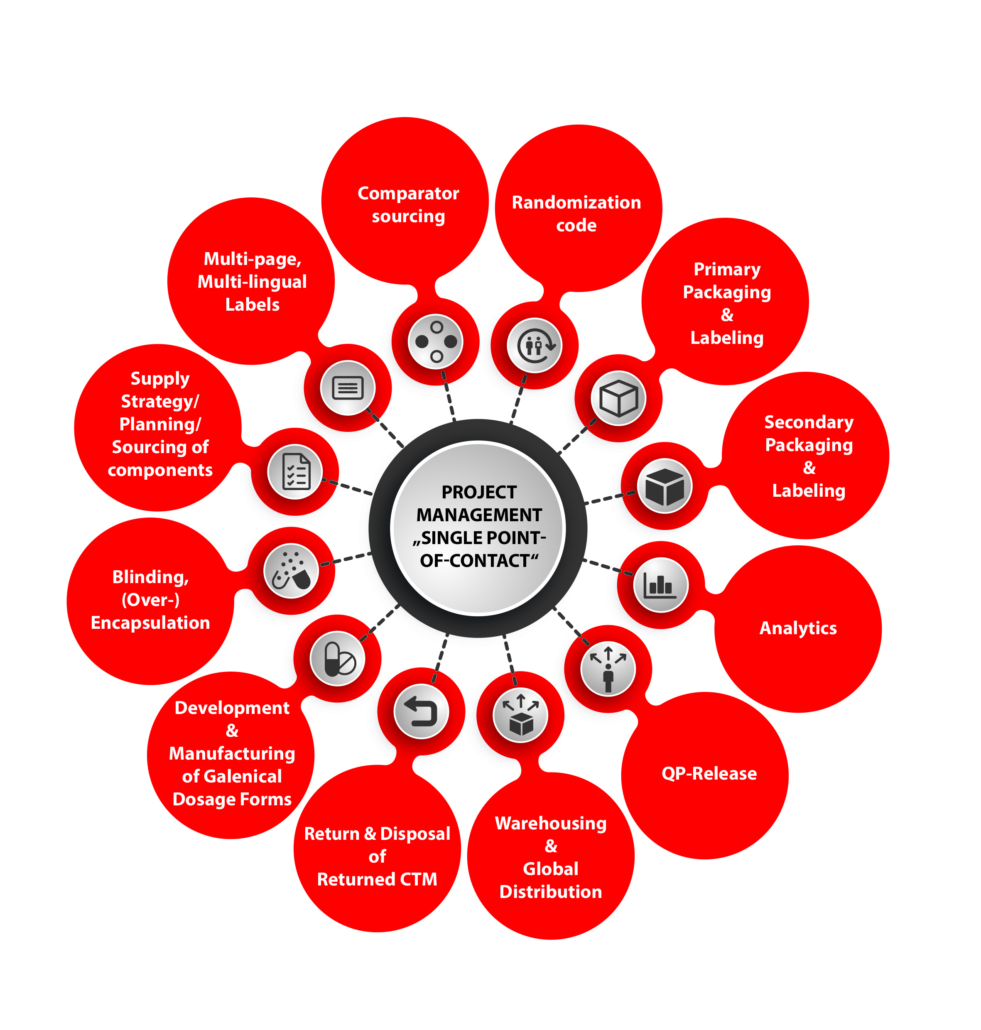
The organisation and handling of clinical supplies is a complex activity. We can offer the following support and expertise
- Comparator sourcing
- Blinding of clinical trial medication
- Primary/Secondary packaging of clinical trial materials
- Storage of the finished units (15–25°C / 2–8°C / -25°C / -80°C)
- Batch Certification and EU Release by EU QP
- Reception of the shipment orders to clinical trial sites incl. triggers from IRT based system
- Picking of the units to be delivered (Patient Kits, Block Boxes, Center Boxes, etc.)
- Shipment preparation incl. packing of passive cooled shipment units with data Loggers
- Commissioning of the carrier along with organizing cold chain management solutions, track & trace, documentation